



Superacid Strength: Triflic acid is one of the strongest acids, categorized as a superacid, enhancing its effectiveness in chemical reactions.
High Stability: The anhydrous form is highly stable, resistant to oxidation and reduction, making it ideal for fine chemical synthesis.
Versatile Applications: It plays a crucial role in synthesizing pharmaceuticals, agricultural chemicals, and polymers.
Non-Oxidizing Nature: Triflic acid’s non-oxidizing properties make it valuable for a variety of chemical processes, especially in pharmaceutical production.
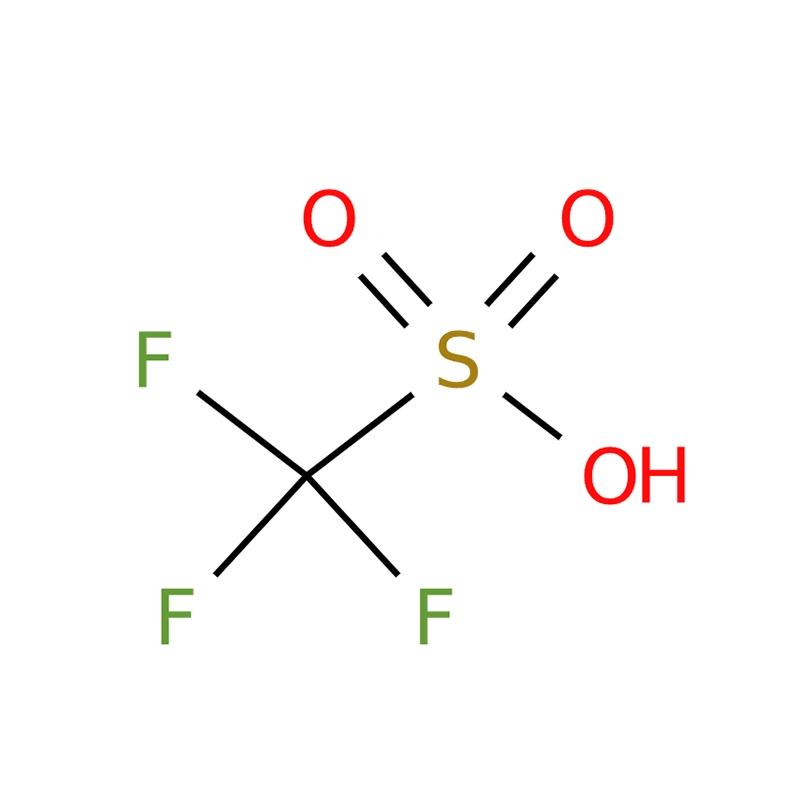
Trifluoromethanesulfonic acid, commonly known as triflic acid (TFMS, TFSA, HOTf, or TfOH), is a highly potent sulfonic acid with the molecular formula CF3SO3H. Renowned for its status as one of the strongest acids, it belongs to a group of compounds referred to as “superacids.” Triflic acid is widely used in the synthesis of pharmaceuticals, agricultural chemicals, and polymers. The anhydrous form, in particular, plays a crucial role in fine chemical synthesis due to its remarkable stability and resistance to both oxidation and reduction.

These properties, combined with its non-oxidizing nature, make triflic acid a valuable tool in various chemical processes. In the pharmaceutical industry, it is integral to the production of several drug classes, including nucleosides, antibiotics, steroids, proteins, and glycosides. Despite its wide applications, triflic anhydride, a derivative of triflic acid, reacts easily with water and is known to have a challenging toxicity profile, limiting its handling in certain contexts.

Parameters
| Melting point | -40 °C |
| Boiling point | 162 °C (lit.) |
| density | 1.696 g/mL at 25 °C (lit.) |
| vapor density | 5.2 (vs air) |
| vapor pressure | 8 mm Hg ( 25 °C) |
| refractive index | n20/D 1.327(lit.) |
| RTECS | PB2771000 |
| Fp | None |
| storage temp | Store below +30°C. |
| solubility | Miscible in H<sub>2</sub>O |
| pka | -14(at 25℃) |
| form | Fuming Liquid |
| color | slightly brown |
| Specific Gravity | 1.696 |
| PH | <1 (H2O) |
| Water Solubility | SOLUBLE |
| Sensitive | Hygroscopic |
| Merck | 14,9676 |
| BRN | 1812100 |
| Stability | Stable. Incompatible with acids, alkalis, metals. |
| InChIKey | ITMCEJHCFYSIIV-UHFFFAOYSA-N |
| LogP | 0.3 at 25℃ |
| CAS DataBase Reference | 1493-13-6(CAS DataBase Reference) |
| NIST Chemistry Reference | CF3SO3H(1493-13-6) |
| EPA Substance Registry System | Methanesulfonic acid, trifluoro- (1493-13-6) |
Safety Information
| Hazard Codes | C |
| Risk Statements | 21/22-35-10 |
| Safety Statements | 26-36/37/39-45 |
| RIDADR | UN 3265 8/PG 2 |
| WGK Germany | 2 |
| F | 3-10 |
| Hazard Note | Corrosive/Hygroscopic |
| TSCA | Yes |
| HazardClass | 8 |
| PackingGroup | II |
| HS Code | 29049020 |
| Toxicity | LD50 orally in Rabbit: 1605 mg/kg LD50 dermal Rat > 2000 mg/kg |

If you are interested in cooperation, please contact us immediately, we will give you feedback as soon as possible!